|
|
Post by ka9q on May 14, 2010 4:00:24 GMT -4
In a traditional chemical rocket, the chemical energy of the propellant is released by burning it. This requires both a fuel and an oxidizer. The problem is that oxygen is heavy and the combustion products of a LOX-LH2 engine have a higher molecular weight than if the reaction mass were hydrogen alone. By eliminating the need to carry an oxidizer and using a propellant consisting of hydrogen only, the exhaust molecular weight is significantly reduced, which results in higher exhaust gas velocity, which results in higher specific impulse. Ah, now I see what you mean. A thermal rocket is a heat engine, and the efficiency with which that engine can convert heat energy to kinetic energy depends on stuff like the molecular weight of the exhaust. Gotcha now. |
|
|
|
Post by thetart on May 14, 2010 8:57:00 GMT -4
In a traditional chemical rocket, the chemical energy of the propellant is released by burning it. This requires both a fuel and an oxidizer. The problem is that oxygen is heavy and the combustion products of a LOX-LH2 engine have a higher molecular weight than if the reaction mass were hydrogen alone. By eliminating the need to carry an oxidizer and using a propellant consisting of hydrogen only, the exhaust molecular weight is significantly reduced, which results in higher exhaust gas velocity, which results in higher specific impulse. Ah, now I see what you mean. A thermal rocket is a heat engine, and the efficiency with which that engine can convert heat energy to kinetic energy depends on stuff like the molecular weight of the exhaust. Gotcha now. But what if the lander is made of balsa-wood? I guess less fuel would be needed but the lander would go on fire, especially in the moons atmosphere. |
|
Bob B.
Bob the Excel Guru?
Posts: 3,072
|
Post by Bob B. on May 14, 2010 9:39:17 GMT -4
Ah, now I see what you mean. A thermal rocket is a heat engine, and the efficiency with which that engine can convert heat energy to kinetic energy depends on stuff like the molecular weight of the exhaust. Gotcha now. I certainly could have explained myself better in my initial post, but sometimes I like to leave my responses somewhat incomplete to see if I can stimulate further discussion. I like it one somebody thinks enough about what I write to ask a follow-up question. An easy way I use to compare one propellant's performance to another is by comparing the value of (Tc/M) 1/2, where Tc is the combustion chamber temperature (approximated by the adiabatic flame temperature) and M is the exhaust gas molecular weight. This relationship comes from the equation for exhaust velocity. It is only an approximation because it ignores differences in specific heat ratio, but it provides a quick check of relative performance. Below are some examples. O/F is the mixture ratio and Tc & M are given at a combustion chamber pressure of 68 atmospheres (1000 PSI): Propellant O/F Tc M (Tc/M)1/2
Nitrogen Tetroxide/UDMH 2.10 3300 21.8 12.3
LOX/Kerosene 2.30 3530 21.6 12.8
LOX/Liquid Hydrogen 5.00 3300 11.8 16.7
Liquid Fluoride/LH2 6.00 3550 10.4 18.5
Liquid Hydrogen (NERVA) ---- 2500 2.0 35.4 |
|
|
|
Post by ka9q on May 14, 2010 9:54:57 GMT -4
For a nuclear thermal engine using LH2 as the propellant, the molecular weight will be 2. How hot would the exhaust have to be to achieve significant disassociation of the H2 into atomic hydrogen and further halve the molecular weight? |
|
|
|
Post by ka9q on May 14, 2010 10:08:52 GMT -4
An easy way I use to compare one propellant's performance to another is by comparing the value of (Tc/M) 1/2, where Tc is the combustion chamber temperature (approximated by the adiabatic flame temperature) and M is the exhaust gas molecular weight. Thanks for the explanation. This is the part of a rocket I've always understood least. I tend to think of a rocket as some sort of power-generating mechanism that linearly accelerates a reaction mass without thinking about exactly how that power gets turned into linear kinetic energy. The non-thermal rockets (electrostatic or electromagnetic accelerators) are a little more intuitive. Unfortunately, it's hard to generate large amounts of electricity without there being a heat engine in there somewhere. And big radiators in space are a pain. Better to use the exhaust itself as your heatsink if you can. I'd read long ago that LH2 was the ideal propellant for a nuclear rocket engine and that it had to do with the molecular weight of the exhaust, but being a little weak in thermodynamics and heat engines (I'm an EE) I never quite understood the connection. Is there a more intuitive way to understand the desirability of lower molecular weight exhausts? I know that one important element in the thermodynamics of heat engines is the number of degrees of freedom in the molecule to absorb energy. A diatomic gas is less preferable to a monatomic gas as it can absorb more energy by spinning or oscillating like a rubber band, while the monatomic gas can't do much but move linearly. But why does lower molecular weight enter into it? |
|
Bob B.
Bob the Excel Guru?
Posts: 3,072
|
Post by Bob B. on May 14, 2010 11:04:39 GMT -4
For a nuclear thermal engine using LH2 as the propellant, the molecular weight will be 2. How hot would the exhaust have to be to achieve significant disassociation of the H2 into atomic hydrogen and further halve the molecular weight? We don't get much dissociation at all until the temperature gets up around 3000 K. The following is the molar fraction of H 2 and H at a pressure of 50 atmospheres. There will be more dissociation at lower pressure and less at higher pressure. Temp H2 H Avg. M
2000K 1.00 0.00 2.02
2500 1.00 0.00 2.01
3000 0.98 0.02 1.99
3500 0.92 0.08 1.94
4000 0.80 0.20 1.81
4500 0.62 0.38 1.63
5000 0.42 0.58 1.43
5500 0.25 0.75 1.26
6000 0.14 0.86 1.15 If we can make a nuclear rocket that can heat hydrogen to 6000K, now we're talking something... (6000/1.15) 1/2 = 72.2. We're probably looking at a specific impulse of about 1800 seconds. (EDIT) I just ran some numbers and I see that my IPS = 1800 s estimate is too high. I forgot that the specific heat ratio of atomic hydrogen is much higher than that of molecular hydrogen. I think an ISP of 1500 to 1600 seconds is more reasonable. Still pretty good. |
|
Bob B.
Bob the Excel Guru?
Posts: 3,072
|
Post by Bob B. on May 14, 2010 11:52:58 GMT -4
I'd read long ago that LH2 was the ideal propellant for a nuclear rocket engine and that it had to do with the molecular weight of the exhaust, but being a little weak in thermodynamics and heat engines (I'm an EE) I never quite understood the connection. I’m a CE myself, so I was never strong at thermodynamics either. When I got interested in rocket engines, however, I was forced to relearn some of it. I still only understand the minimum needed to get by. Is there a more intuitive way to understand the desirability of lower molecular weight exhausts? I know that one important element in the thermodynamics of heat engines is the number of degrees of freedom in the molecule to absorb energy. A diatomic gas is less preferable to a monatomic gas as it can absorb more energy by spinning or oscillating like a rubber band, while the monatomic gas can't do much but move linearly. But why does lower molecular weight enter into it? This is the way I’ve always thought about it (right or wrong)… What’s happening in the nozzle is the high-pressure, high-temperature, low-velocity gas in the combustion chamber is transformed into lower-pressure, lower-temperature, higher-velocity gas at the nozzle exit. The thermal and potential energy of the gas is transformed into kinetic energy. To carry that kinetic energy, a lower mass-molecule must travel faster. Kinetic energy is KE = mv 2/2. In a rocket engine, the kinetic energy comes from the transformation of the thermal energy released during combustion; therefore, we can equate KE to the combustion temperature, Tc. We now have Tc = mv 2/2, or v = (2*Tc/m) 1/2. Any reactant that produces a lot of energy when burned and results in low-mass products makes the best chemical rocket fuel. |
|
Bob B.
Bob the Excel Guru?
Posts: 3,072
|
Post by Bob B. on May 15, 2010 14:37:32 GMT -4
I just did a math experiment that seems to confirm what I suspected. Let's assume that as a gas cools its heat energy is transformed into kinetic energy. The amount of kinetic energy should be the difference between the enthalpy (heat content) of the gas at the higher temperature and that at the lower temperature. Knowing the kinetic energy and the mass of the gas, we can calculate the gas velocity. I found the enthalpy, H, of the four most common exhaust gases at temperatures 3000 K and 1000 K. I then took the difference and calculated the velocity from KE = mv 2/2. Here are the results: Gas Mol.Wt H @ 3000K H @ 1000K Delta-H Velocity
J/kg J/kg J/kg m/s
H2 2 4.4020E+7 1.0261E+7 3.3759E+7 8,217
H2O 18 -6.4091E+6 -1.1981E+7 5.5719E+6 3,338
CO 28 -6.0690E+5 -3.1722E+6 2.5653E+6 2,265
N2 28 3.3105E+6 7.6605E+5 2.5445E+6 2,256 The goings on inside a rocket engine is more complex than this, but I think this clearly shows that low molecular weight gases will be propelled to higher velocities when expanded and cooled. |
|
Jason
Pluto
May all your hits be crits
Posts: 5,579
|
Post by Jason on May 17, 2010 11:46:00 GMT -4
Is it always the case that a low-density gas is the most efficient?
A heavier gas accelerated to the same speeds would presumably provide more thrust than a lighter gas, but I can understand how it may be less efficient to accelerate it.
|
|
Bob B.
Bob the Excel Guru?
Posts: 3,072
|
Post by Bob B. on May 17, 2010 15:51:41 GMT -4
Is it always the case that a low-density gas is the most efficient? I think you mean low-molecular weight, not low-density. Those terms don't mean the same thing, though at a given temperature and pressure, the lower molecular weight gas will also have the lower density. All other things being equal, such as temperature and pressure, a low-molecular weight gas will always produce a higher exhaust velocity than a high-molecular weight gas. This can be seen by examining the exhaust velocity equation: 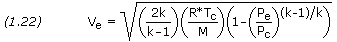 A heavier gas accelerated to the same speeds would presumably provide more thrust than a lighter gas, but I can understand how it may be less efficient to accelerate it. There are other factors, in addition to molecular weight, that contribute to an engine's exhaust velocity, such as combustion chamber pressure and nozzle expansion ratio. However, all other things being equal, you'll get a greater velocity with a lighter propellant. Thrust equates to the momentum of the exhaust gas, where momentum is Mass X Velocity. To get more thrust you must either eject more mass or increase the ejection velocity. For a given propellant, there are things that can be done to increase velocity, such as operating at a higher pressure, but there are practical limitations. You reach a point where the penalty is larger than the margin of return. If you want a much greater velocity, you have to consider low-molecular weight propellants. The only real option is liquid hydrogen because it's the only fuel that gives a great enough increase in performance to warrant dealing with its drawbacks (cryogenic and low density). A fuel such a liquid methane gives better performance than, say, kerosene, but not that much better. If we're going to introduce the problems of dealing with cryogenic methane, we might as well use LH2 and get a much better performance. There are also propellants such as fluorine/LH2 that could yield a performance even better than LOX/LH2, but fluorine is so toxic and reactive that it is a nightmare to deal with, making it not worth the trouble. It should be noted that a low-molecular weight gas will possess more momentum per kilogram, however less momentum per mole. |
|
Jason
Pluto
May all your hits be crits
Posts: 5,579
|
Post by Jason on May 17, 2010 17:39:48 GMT -4
I think you mean low-molecular weight, not low-density. Those terms don't mean the same thing, though at a given temperature and pressure, the lower molecular weight gas will also have the lower density. Yes, that is what I meant. Which is what I thought. For a given ejection velocity, ejecting more mass would produce more thrust, right? Then wouldn't a higher-density propellant produce higher thrust at a given velocity? And what about using some acceleration method other than burning/exploding it, such as a magnetic accelerator throwing chunks of iron out the back of your ship rather than burning gas? |
|
Bob B.
Bob the Excel Guru?
Posts: 3,072
|
Post by Bob B. on May 17, 2010 21:49:58 GMT -4
For a given ejection velocity, ejecting more mass would produce more thrust, right? Correct. Then wouldn't a higher-density propellant produce higher thrust at a given velocity? That depends on what you mean. At a given velocity one mole of a heavy gas will produce more thrust than one mole of a light gas. However, one kilogram of gas ejected at a given velocity will produce a given amount of thrust regardless of the molecular weight. That is, if we expel gas at a rate of one kg/s at a velocity of 3,000 m/s, we'll get 3,000 N of thrust -- it doesn't matter whether the gas is hydrogen, water vapor, carbon monoxide, or something else. And what about using some acceleration method other than burning/exploding it, such as a magnetic accelerator throwing chunks of iron out the back of your ship rather than burning gas? What you describe is the principle behind ion propulsion. An ion thruster uses electricity to accelerate charged ions (usually Xenon) and expel them at very high velocity, resulting in high specific impulse. The good thing is that you need very little propellant; the bad thing is you need vast amounts of electricity. Since we're limited by the amount of electricity that can be reasonable produced, ion propulsion is practical only in small low-thrust motors. There are also other forms of electric propulsion, though I'm not very familiar with them. I don't know enough to comment on the practicality of using a heavy material like iron as a reaction mass. My gut feeling is that it would be easier to electrically accelerate a lightweight ion than it would be to magnetically accelerate a heavy iron molecule. Ka9q is the electrical engineer, perhaps he's more qualified to comment than I am. |
|
|
|
Post by laurel on May 17, 2010 22:13:40 GMT -4
Do you understand why this sort of claim isn't convincing? You're not offering evidence for any alternative scenario at all, let alone one as well-documented as Apollo. And I'd really like you to explain the contradictory information about Van Allen radiation that you posted in your blog.
I'll come to that soon.Seven days later and no answer. This must be a new definition of "soon" that I'm not aware of. I can't help contrasting this with what happened when I e-mailed Eric Jones with a question about Apollo 14. He answered the question the next day. Hagbard Celine wants us to be suspicious of NASA and its associates, but he is the one acting evasive. |
|
|
|
Post by ka9q on May 19, 2010 12:29:18 GMT -4
For a given ejection velocity, ejecting more mass would produce more thrust, right? Then wouldn't a higher-density propellant produce higher thrust at a given velocity? Yes, but it also means a heavier load of propellant at launch, which means more thrust to get it off the ground. Specific impulse is a measure of the impulse (force times time) for each unit of propellant mass. It's understood that you can make the impulse as big as you want by carrying more propellant. |
|
|
|
Post by ka9q on May 19, 2010 12:41:59 GMT -4
The good thing is that you need very little propellant; the bad thing is you need vast amounts of electricity. Right. And there does exist a practical means of generating a very large amount of power in small weight and volume: a nuclear reactor. But its power comes out as heat. It has to be converted to electricity before it can be used in an ion engine. This is done with a heat engine; large power plants on earth traditionally use the Rankine cycle. Because it uses coolant in a closed loop, it is likely to be the only practical means in space as well. But this introduces a drawback. Recovering and recirculating the reactor coolant after it has done work in a turbine requires that you dump its remaining heat. On earth we use large bodies of water or water evaporation in cooling towers for this purpose, but in space we don't have this option. We'd have to use radiators, and depending on how much heat they have to radiate and especially the resulting temperature of the recovered coolant, they can become seriously large. Certain kinds of missions can still justify this complexity, but in general it would be very hard to beat the simplicity and high thrust levels you can get by using a rocket as the heat engine for converting reactor heat into thrust. Since the propellant/coolant flows through the reactor and out the nozzle without being recovered, we don't need radiators and the thrust can be quite high. |
|